EU8. Energy for Heating and Cooling

Chapter 8
I. Controlling the Flow of Heat
Keeping comfortable, or simply staying alive, requires a temperature that’s not too hot and not too cold. As warm-blooded creatures, we have a built-in system for heat control. As intelligent humans, we augment our built-in systems with technologies—clothing, housing, and fire—that enable us to live in places where we could not otherwise survive. We have also learned to use heat in new ways—to make ceramics, to refine metals, to power automobiles and factories, and to fire rockets into space.
To conserve energy, we need to be able to control the flow of heat.
The science of heat energy has its own name—thermodynamics. To keep the heat we use in the places we want it, and out of the places we do not want it, we must understand the three processes of heat flow: conduction, convection, and radiation.
I-A. Heat Flow By Conduction
Your most vivid experiences of heat moving has probably been when you directly touch something. Climb into a car that has been sitting in the hot sun. The upholstery is toasty hot from hours of heating by the greenhouse effect. Ouch! How about the way your mouth feels after you bite off a big chunk of popsicle? In both cases your sensations are an immediate result of heat moving by conduction.
Conduction is movement of heat that occurs when two objects are in direct contact with each other.
One principle of thermodynamics is that heat flow by conduction goes from the hotter object to the cooler one. When you bite a popsicle, you are sensing the flow of heat from the inside of your mouth into the chunk of popsicle. When you sit on a hot car seat, your skin is sensing the flow of heat from the car seat into your body.
Put your hand on a page of a book, then the floor, a window, a rug, something metal, and something plastic. Do they all feel the same temperature? They may be the same temperature, even if they do not feel the same temperature. How can this be? The reason is in how well they conduct heat. Metal conducts heat very well so when you touch it at room temperature, heat flows quickly out of your body—you feel cold. Something made of wood, also at room temperature, feels warmer because heat flows very slowly out of your body into the wood, which is a poor conductor of heat.
Question 8.1
What are other examples of heat conduction

EU8.1. Investigation: How Does a Thermos Bottle Know?
Three science teachers were relaxing after a hard day at school. Their talk turned to the great technological achievements of the modern world. One said, “There is nothing that can beat computers. Computers can do more and more, and they are getting smaller and smaller, and cheaper and cheaper. It’s amazing!”
“What about lasers?” the second teacher said. “They are everywhere! I’ll bet in a few years lasers will be used in ways we cannot even imagine now. Lasers certainly rival computers!”
“And then there are Thermos bottles,” added the third teacher.
“Thermos bottles?” said the others. “What is so special about Thermos bottles?” “Well,” explained teacher number three, “if you put something hot in one it stays hot. If you put something cold in one it stays cold. How does the Thermos bottle know what it’s supposed to do?”
As you read this chapter, and you discover other ideas of how a Thermos™ bottle works, feel free to change your explanation
Question 8.2 How does the Thermos™ bottle know? Write down your ideas about how you think a Thermos™ bottle keeps hot things from cooling off and cold things from warming up.
Insulation—Preventing Heat Loss By Conduction
In the walls or ceiling of a house or apartment, insulation helps minimize house heating costs and energy waste by keeping heat from escaping. In 2013, 40% of total U.S. energy consumption was consumed in residential and commercial buildings, or about 40 quadrillion British thermal units. [from U.S. Energy Information Administration. The British thermal unit (BTU or Btu) is the amount of energy (heat) needed to cool or heat one pound of water by one degree Fahrenheit.] Heating and cooling are generally the biggest energy uses in buildings.
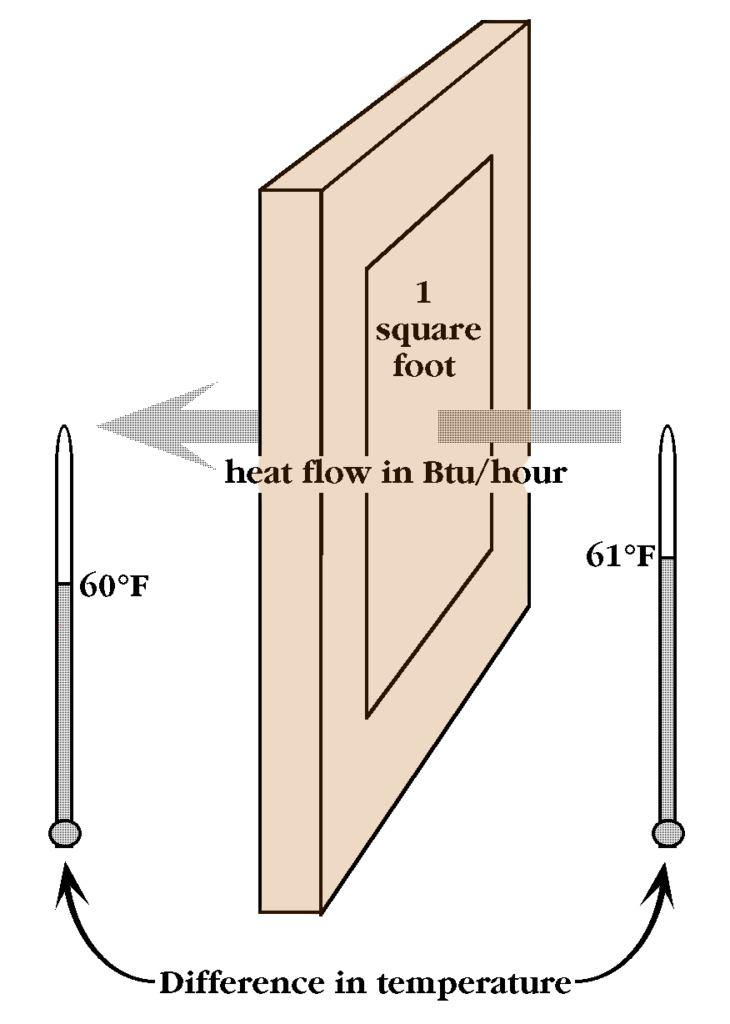
Insulation conducts heat poorly. The measure of a material’s ability to resist the flow of heat is called its R-value. A standard single pane window has an R-value much less than one. A lot of heat escapes through a single pane window. In cold climates, it is wise for home owners to insulate their ceilings to at least R-30.
R-value is the difference in temperature on either side of a material, divided by the rate of heat flow in Btus per hour per square foot.
For example, a window with an area of one square foot, that is one degree Fahrenheit warmer on one side, lets heat flow through at a rate of 1 Btu per hour. It would be R-2 if it allows only one-half a Btu to flow through per hour; and R-30 if just one thirtieth of a Btu flows through per hour.
A home builder in a cold climate is always interested in the best insulation. Goose down—tiny feathers used in expensive pillows and sleeping bags—has a very high R-value. But insulating a house with goose down would not be very practical. For a building, the choice of insulation depends not only on R-value but also on cost, strength, durability, ease of handling, and fire safety. Materials used for insulation include fiberglass, foam boards, sheet rock, double pane windows, and wood. Some insulation is even made from cattle hair or sugar cane fiber.
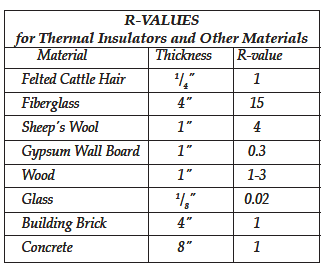
There is a wide range of R-values in common building materials. A four-inch thick brick wall is not a good insulator. By making it twice as thick you could double its ability to reduce heat flow, but you could get the same insulation by adding a layer of felted cattle hair. It would be less expensive and it would take up less space.

EU8.2. Investigation: Compute an R-Value
- Find the temperature difference T on the two sides of the material
- Multiply it by the area A of the material
- Divide it by the rate of heat flow H through the material.
R-Value = T x A / H
Question 8.3
What is the R-value of a 2-square-foot window that allows 20 btu of heat to flow through it per hour?
The indoor temperature is 65°F and the outside temperature is 55°F.
Celsius (°C) to Fahrenheit (°F)*:
Use this formula to convert °C to °F:
°F = (°C x 9/5) = 32
We use non-metric temperature (°F) in our calculations because most American energy-related industries have not converted to the metric system. To use °C would add an extra step of complexity to figure out the R-value of insulation, which use btus and °F.
I-B. Heat Flow By Convection
Air conducts heat very poorly. Yet, when a house is built in a cold climate the air spaces in the walls and the ceilings are often filled with insulating materials. Why isn’t the air alone enough to do the job? The answer is that air does not sit still, and when air flows, its energy flows with it.
The flow of heat by way of a moving fluid is called convection.
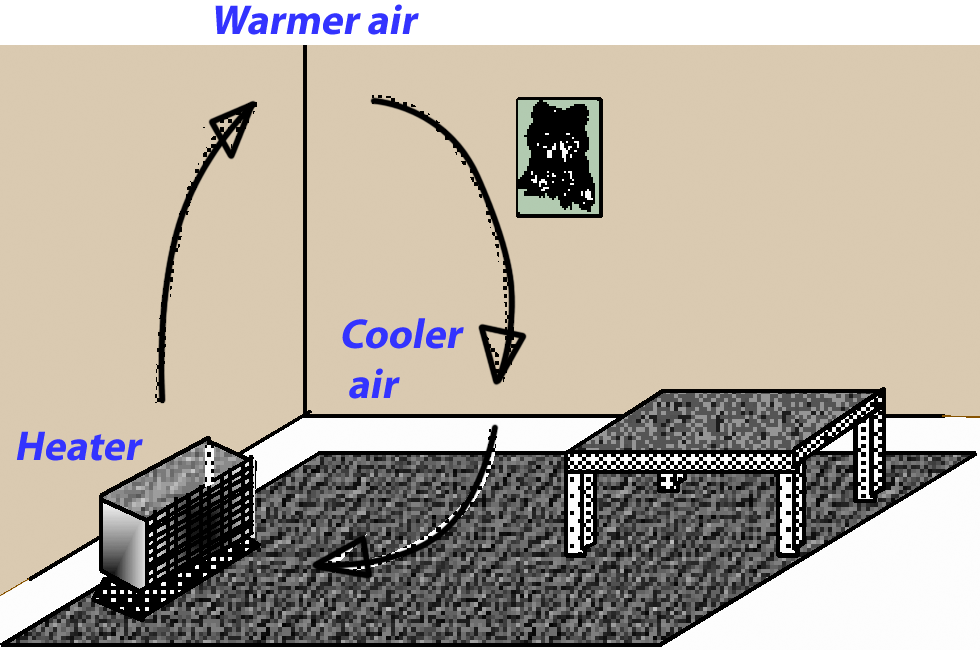
As air warms up, it expands and becomes less dense than the surrounding cool air. The warm (less dense) air is pushed upwards by the cold (denser) air. That principle is what makes a hot air balloon float upwards. A heater in a room warms the air just above it. The air rises to the ceiling, cools and drops to the floor, where it is heated again. That is a convection current.
Convection currents do not always distribute heat where it is needed. A layer of hot air near the ceiling does not give much comfort to the people below. A ceiling fan can send the warm air back down. Although a fan uses electricity, it may save on the overall cost of energy bills.
A fire in the fireplace might be a good way to warm the place up, right? Wrong! Convection strikes again, in a big way. Convection does not just carry smoke up the chimney; it carries warm air up the chimney too! An airtight wood stove works better by limiting convection.
Convection is one reason it is especially important to insulate a roof well. If it is 60°F inside and 50°F outside (a 10°F difference), it is possible that convection has carried a layer of warm air to the roof raising the inside temperature to 80°F at the roof (a 30°F difference). If the R-value were the same in the walls and the ceiling, the heat loss per square foot through ceiling would be three times the heat loss through the wall
I-C. Radiant Energy—Heat Flow at the Speed of Light
What material would stop heat flow best of all? The best heat barrier is nothing—a vacuum—empty space. But even a vacuum cannot stop heat completely. If it could, we would never be able to feel the warmth of the Sun across the emptiness of space. Some heat energy passes between objects that are not touching, even if there is empty space between them. That process of heat flow is called radiation; and the kind of energy that travels by radiation is called radiant energy (or electromagnetic energy).
Heat is always being converted into radiant energy. You can see (and feel) this transformation every time you use a incandescent light bulb. Inside the light bulb is a tungsten metal filament—the long narrow wire—which heats up to more than 1832°F as electricity flows through it. Some of the heat changes into brilliant white light—a visible form of radiant energy.
Look around you now. There is probably nothing near you that is as hot as the inside of a light bulb, but everything around you has some heat in it. Everything around you is giving off some of that heat as radiant energy. Only the very hot things such as light bulbs give off light you can see. Cooler objects give off infrared energy that you cannot see.
Objects lose heat when they give off radiation, and they heat up when they absorb radiation.
Which gets hotter in the sunshine; a white concrete sidewalk, or a black asphalt road?
You certainly know the answer if you have ever stepped off the sidewalk to cross the street in bare feet on a very sunny afternoon. Dark surfaces absorb visible light better than light surfaces. The absorbed energy becomes heat. Most objects, no matter how dark or light they look, absorb infrared radiation, so heat lamps are made to emit infrared energy.
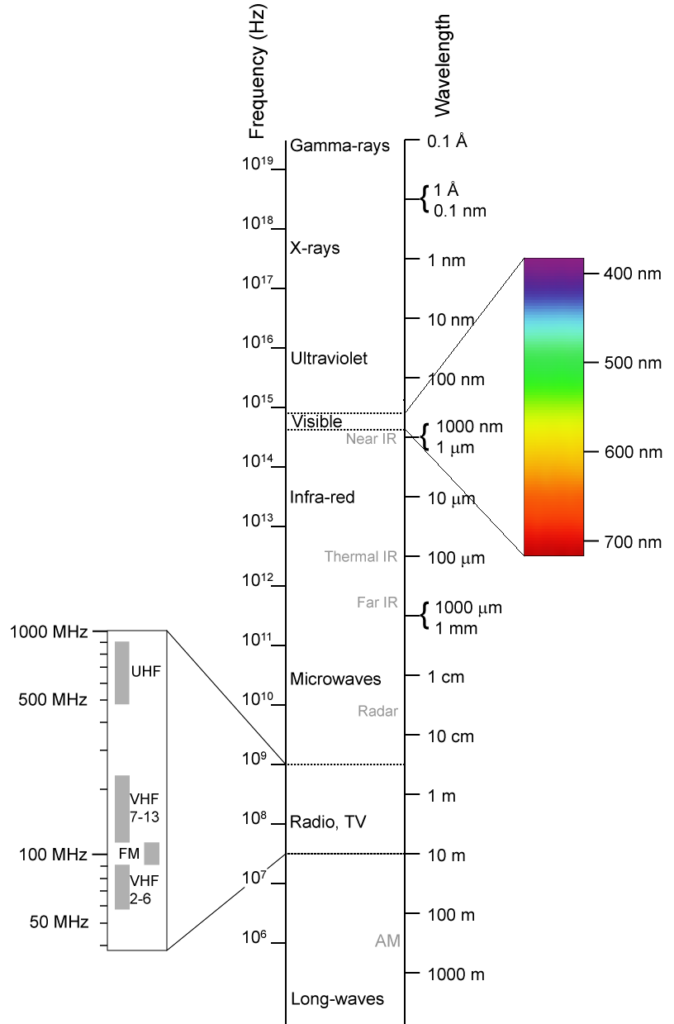
The Electromagnetic Spectrum
Traveling waves of electric and magnetic fields are known as electromagnetic radiation. Like water waves which can be long rolling swells or short choppy waves, electromagnetic waves can be long or short. The length of the wave—called the wavelength—determines how it behaves.
Visible light has wavelengths between 400 and 700 nanometers. (A nanometer is one billionth of a meter.) The exact wavelength determines the color of the light.
Visible light is partly absorbed and partly reflected by most materials. We see things by the light they reflect. Objects are warmed by the light they absorb. Infrared radiation, which has longer wavelengths than visible light, is easily absorbed by most materials and will heat them up. Ultraviolet light (UV) has shorter wavelengths than visible light. UV light can heat an object, but it can also cause chemical changes and damage to materials including skin.
Near one end of the electromagnetic spectrum is radiation with the longest wavelengths (microwaves and radio waves), while at the other end is radiation with the shortest wavelengths (X-rays and gamma rays).
Question 8.4
What personal experiences have you had that involve a particular portion of the electromagnetic spectrum?
II. Same House, Different Seasons
The column in the right side of the house in the diagrams below represent a “thermal storage wall” that is intended to absorb heat during the day to store it for release inside the house during the night.
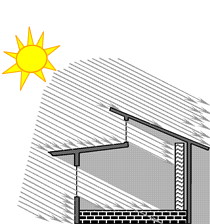
In WINTER the sun’s rays shine in
and are abosorbed by the thermal mass
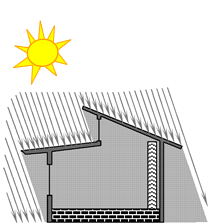
In SUMMER the sun’s rays cannot
shine into the house
Question 8.5
How does the function of the thermal storage wall change over the four seasons?
III. Passive And Active Solar House Heating
If your home is heated by burning natural gas, the heat is probably spread around to the living spaces in the house by blowing hot air through ducts or pumping hot water to radiators. In solar heated buildings, heat can come into the living spaces directly through the windows, or accumulated in solar collectors using either hot air or hot water for collecting heat. The heat is stored in rock bins (with hot air solar collectors) or water tanks (with solar water heating panels). In passive solar houses, the collected heat is distributed through the house by convection. If fans or pumps are used to circulate the heat through the living spaces, then the building design is active solar.
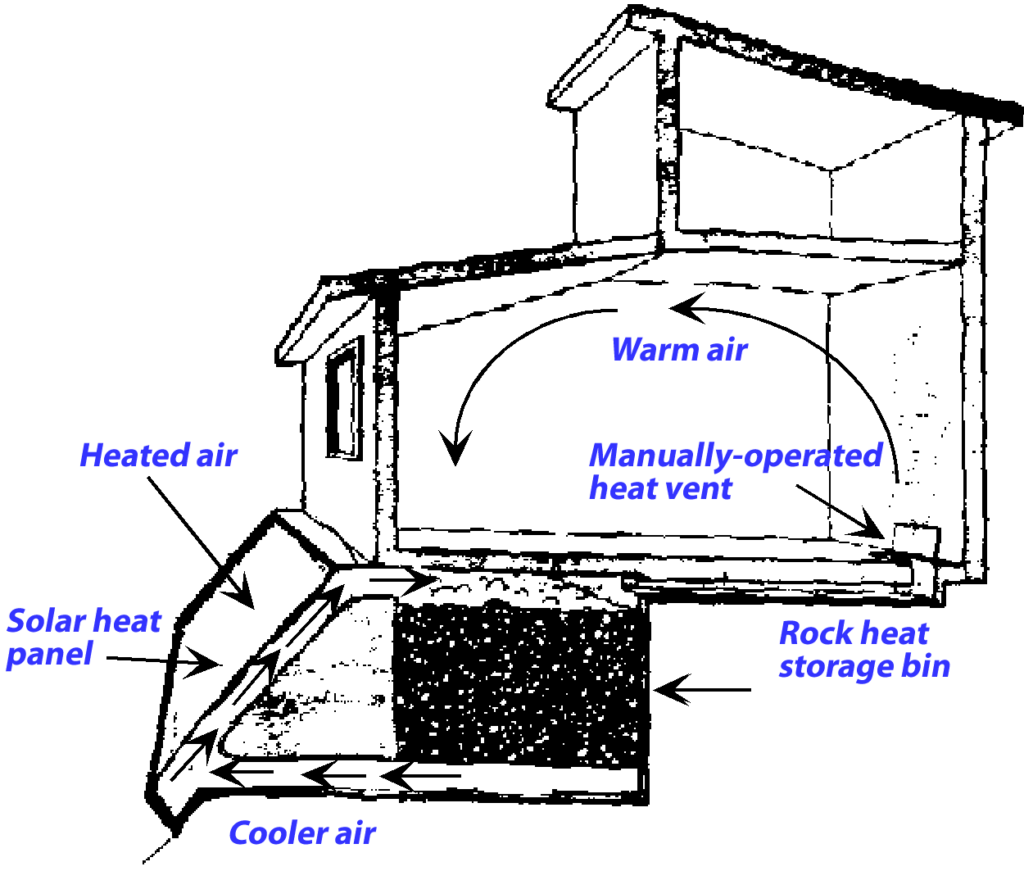
Passive Solar House Design
Hot air from a rock heat storage bin is distributed by natural convection through the living areas of the house.
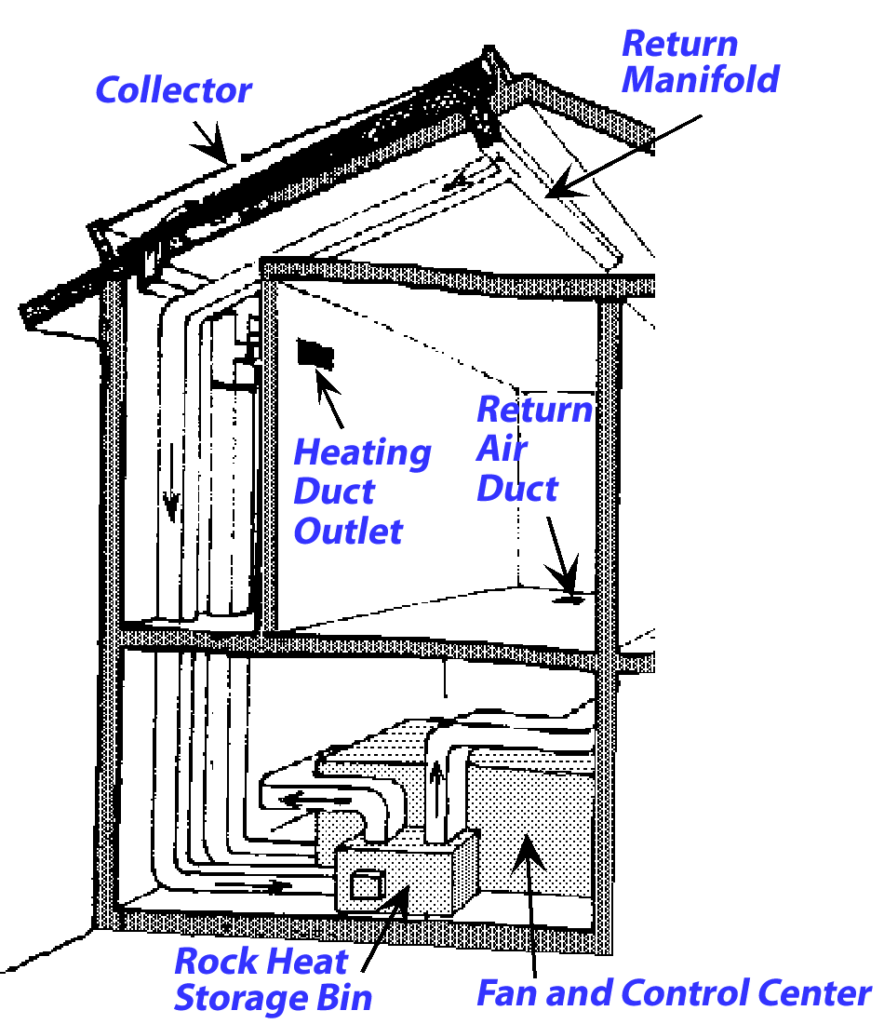
Active Solar House Design
The fluid that distributes heat in buildings with active solar heating can be either air or water. Pictured here is a hot air system with a rock bin for heat storage and fans for distribution of heat.
Question 8.6
What is the same in both a passively and actively solar heated home? What is different?

How to Cool a House Without A/C
According to the U.S. Census Bureau, there are approximately 124 million households in the U.S.A. Data from the EIA tells us that 90% of these households have some form of air conditioning.
What does your math tell you how many households are without air conditioning?
If you do not have air conditioning or know people who do not have air conditioning, look at this article about how to cool a house without air conditioning.
https://todayshomeowner.com/hvac/guides/how-to-cool-a-house-without-ac/
- Which ideas would be effective and appropriate for you to use to keep things cool without air conditioning?
- Which ideas might help conserve energy even for households that have air conditioning?
IV. Solar Hot Water
It’s easy to heat water with sunlight. Solar heated water can be stored for home heating, or used for bathing, laundry, or dishes.
Here is a diagram showing a typical solar water heating panel that can be installed on the roof of a house.
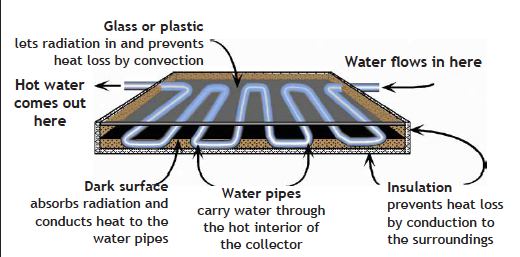

EU8.3. Investigation: Refrigerators
Josh and Bert met after school and were looking for a snack in the kitchen at Bert’s house. Josh said, “This kitchen is hot! Can’t you open a window or something?”
“What good would that do?” Bert said. “It’s even hotter outside. Besides, we shouldn’t open the window because the air conditioning is on.”
Josh said, “Let’s open the refrigerator door. That should cool things down.” Bert pulled open the refrigerator and they both stood in front of it and enjoyed the feeling of the cool air.
Bert said, “I guess no food will spoil if we leave the refrigerator open for a few minutes. Listen to it hum!”
“You know,” said Josh, “our science book says that energy is never used up. It just changes from one kind of energy to another. So where does the energy go that runs the refrigerator?”
“The refrigerator takes electrical energy and uses it to make things cold. So I guess it turns electrical energy into cold energy,” Bert said.
“No, that can’t be right,” Josh said. “I know that heat is a kind of energy, but I’ve never heard of cold energy.”
In charged Bert’s mother. “Close that refrigerator!” she said. “It’s too hot in here already!”
Does Bart’s mother’s request make sense?
Josh knew that energy is never used up. Bert was aware that heat is a form of energy. They have an inkling of the first law of thermodynamics: (also called the law of conservation of energy) that energy can neither be created nor destroyed, but can change from one form to another. The second law of thermodynamics, that everything tends to go from a state of order to disorder, is harder to grasp. But one of its consequences is that heat flows from hotter places to cooler places. A refrigerator, however, seems to do just the opposite. It makes itself cold on the inside by moving heat from the cooler inside to the warmer outside.
The second law of thermodynamics does allow for heat to move from a cooler place to a hotter place, but it always requires work—the input of more energy. The energy that does the work to move the heat also adds to the total amount of heat in the system.
Bert’s mother understood that by forcing the refrigerator to keep running, the boys were producing even more heat in the kitchen.
V. How a Refrigerator Works
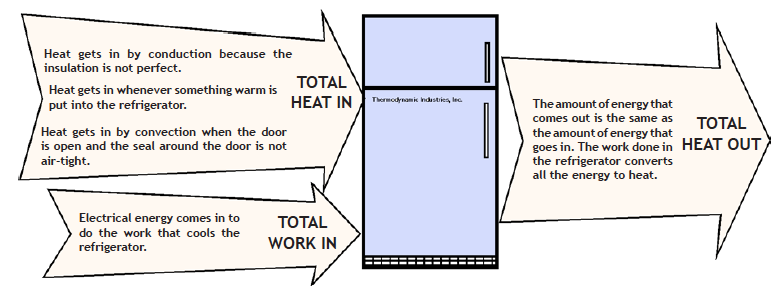
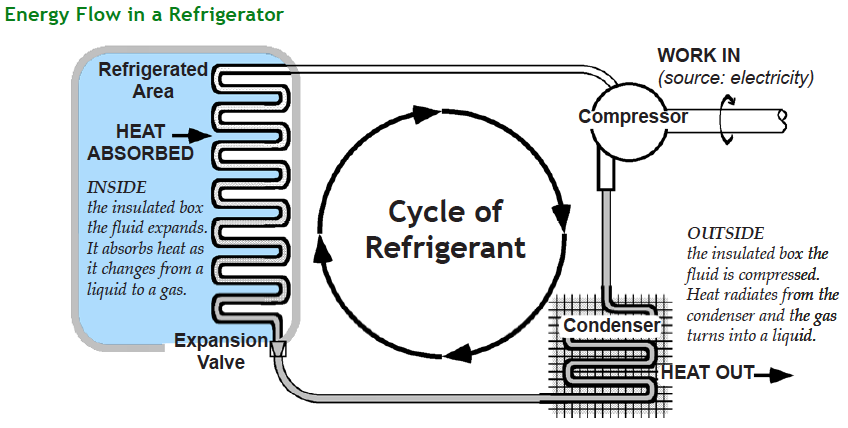
A refrigerator is a double-walled insulated box with a system of pipes between the walls that have a “working fluid” flowing through them.
When the fluid runs through the pipes inside the insulated box, it changes from a liquid to a gas as it absorbs heat from inside the box. The fluid is then compressed and pumped outside the insulated box to condenser coils where it changes back to a liquid by releasing heat it absorbed from inside the box. The condenser radiates heat to the surrounding air outside the refrigerator.
Building A Better Refrigerator
In 1993, a consortium of 24 utility companies sponsored a contest with a $30 million prize for the best new refrigerator on the market. Entrants had to build a product 50% more energy efficient than current refrigerators. They also had to have a plan to market their product.
Another requirement for the winning refrigerator was that it could use no chlorofluorocarbons—CFCs. For decades the working fluid in most refrigerators has been freon, a CFC fluid. Freon released from air conditioners and refrigerators is a major contributor to the destruction of ozone in the upper atmosphere that protects us from the lethal ultraviolet rays of the Sun. It has become urgent that freon in old refrigerators be recycled rather than released to the air and that alternatives to freon be used as working refrigerator fluids. Whirlpool won that contest but you can find energy efficient refrigerators among all the major brands.

EU8.4. Investigation:
Exploring A Refrigerator
Let’s explore some parts of the refrigerator that keep it cold and some parts that move heat energy from the interior to the exterior of the refrigerator.
Materials
- Refrigerator or freezer.
- Refrigerator owner’s manual if it is available.Examine the walls and door of the refrigerator.
Look for features that help keep heat out. Look on the outside of your refrigerator for the condenser coils. The owner’s manual may help you to find them. They are probably on the back side or the bottom of the refrigerator, or both. Look for features that help send heat from the coils into the air away from the refrigerator.• Did you find the condenser coil? • Did it feel warm? • What color is it? • How does its color help it to function properly? • Why do you think that this part of the refrigerator has so many thin pieces of metal welded to it? If you check your refrigerator’s manual it will almost certainly give instructions on cleaning this part of the refrigerator.
Question 8.7
How might a dusty condenser coil keep heat from getting away from your refrigerator?
Question 8.8
Does this article from 1991 have any tips you could follow today? Why does an electric company care if your refrigerator is energy efficient?
For more up to date information about cutting refrigeration costs, visit https://www.energystar.gov/productfinder/product/certified-residential-refrigerators/results

